 
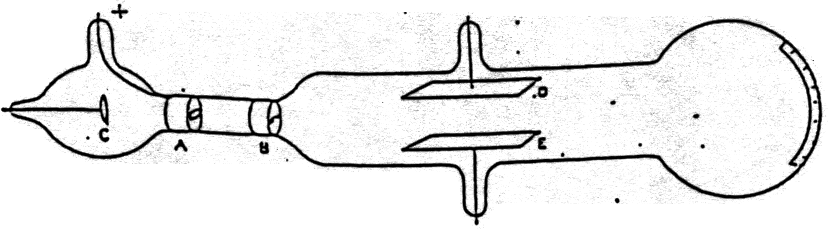
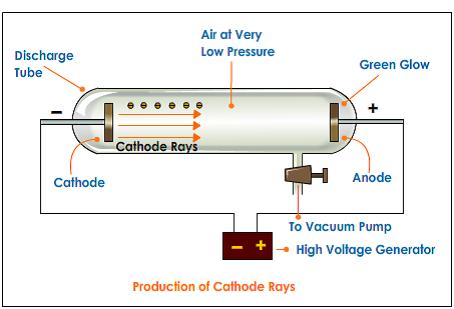
His model can be compared to a plum pudding or a watermelon.Therefore, the atom has no real charge and is electrically neutral. The positive charge and the negative charge are comparable in magnitude.An atom approximates a sphere of positive charge and negative charge existing inside the sphere.The atom is the minimal unit of matter that can take part in a chemical reaction.Įnglish physicist JJ Thomson provided one of the first atomic prototypes comprising the newly discovered particles.Atoms of similar elements can converge in more than one ratio to cast two or more compounds.Atoms of dissimilar elements may converge in a definite, simple and whole number ratio to cast compound atoms.Atoms can neither be developed nor destroyed.Atoms of a similar element are the same shape and mass but diverge from the atoms of other elements.All matter comprises indivisible particles called atoms.Postulates of Dalton’s atomic theory are: His discoveries were based on experiments and the laws of chemical combinations. Dalton’s Atomic TheoryĪ British schoolteacher named John Dalton came up with his theory on atoms in 1808. The images may be electrical waveforms (oscilloscope), pictures (TVs, computer monitors), sonar targets, or other forms. The beams from the guns are manipulated to produce images on a glowing screen. What is the wavelength (in mm ) of a particle of mass 6.62×10−29g moving with a velocity of 103ms−1?.The cathode ray tube, also known as the vacuum tube, contains one or more electron guns.Which of the following is the correct unit of angular momentum of an lectron in an orbital of an atom?.The line of longest wavelength corresponds to n2=3.The energy of an electron in the 3S orbital (excited state) of H - atom is.Among the elements from atomic number 11 to 36, the number of elements which have an unpaired electron in their s subshell is.If the energies of the two photons are in the ratio of 3 : 2, their wavelengths will be in the ratio of.Electronic configuration of calcium atom can be written as.Uncertainty in position of an electron (mass of an electron is =9.1×10−28g)….4d, 5p, 5f and 6p orbitals are arranged in the order of decreasing energy.According to the Bohr Theory, which of the following transitions in the hydrogen atom will give rise to the least energetic photon?. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
